Medical Device Quality Management System (QMS).
Certified to ISO 13485:2016 Standards. This is not a general ISO 9001 business license; it is a regulatory mandate for medical manufacturing.
Audit Note
AUDIT NOTE: Ensure your supplier holds ISO 13485, not just ISO 9001. Only 13485 covers 'Risk Management' required by MDR/FDA.
The 'Medical-Grade' Distinction
| Feature | ISO 9001 (General) | ISO 13485 (ValidTi) |
|---|---|---|
| Focus | Customer Satisfaction | Patient Safety & Efficacy |
| Risk Management | Optional | Mandatory (ISO 14971) |
| Traceability | Basic | Full Batch History Record |
| Cleanliness | Standard | Contamination Control |
Loss of Traceability (Recall Risk)
Risk Addressed
Loss of Traceability (Recall Risk)
In the event of a clinical failure, the 'Paper Trail' is your only defense. ValidTi's QMS enforces a strict 'Linkage Protocol': Every disc is laser-marked with a Lot Number that links back to the Vacuum Arc Remelting (VAR) log and the Chemistry Certificate. No anonymous metal.
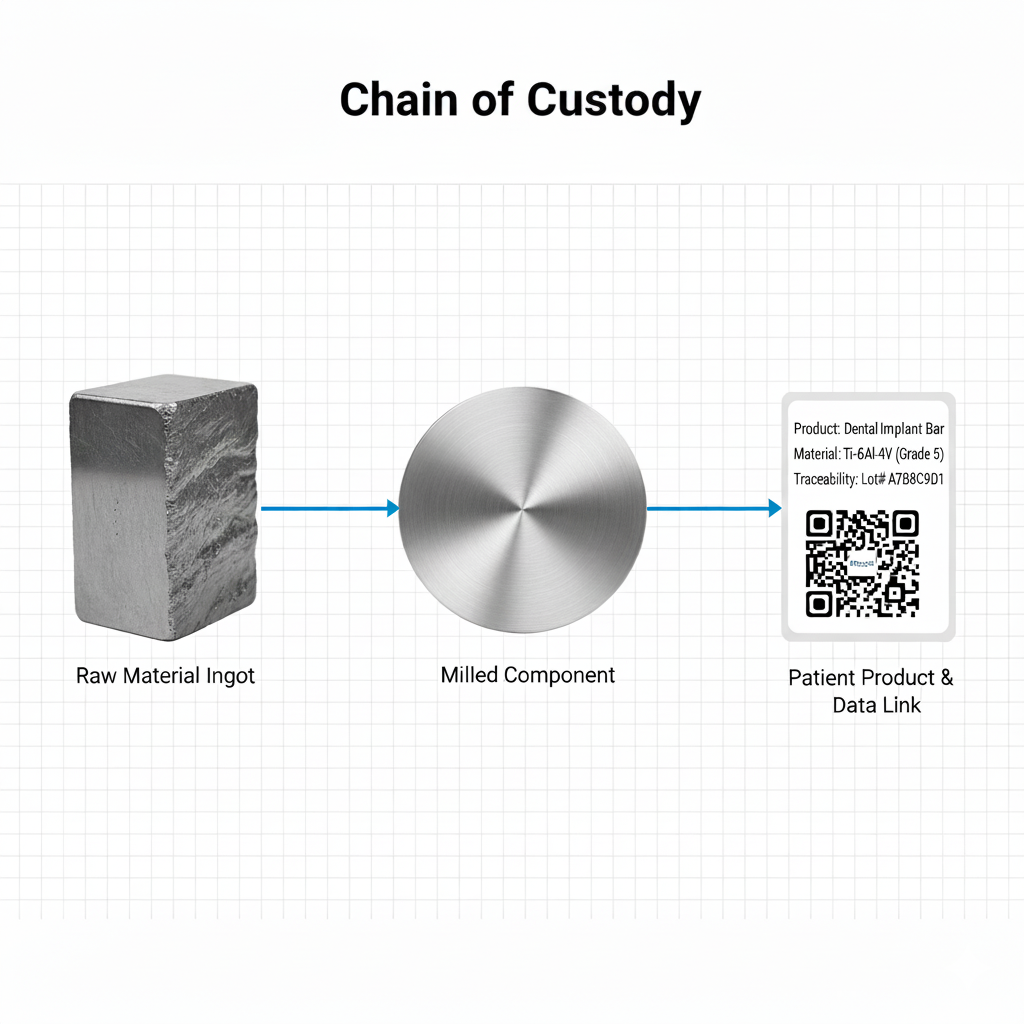
Fig 1. End-to-End Traceability Chain. Flowchart showing data thread from ingot to patient.
Quality Management Documentation / Audit Vault
Secure Audit Request Portal // QMS Revision: 2025-Q1